Document Control for Compliance
Document control software is essential for quality and compliance. Quality managers and document controllers have an effective way to manage policies and procedures, record log files, process change requests, and capture required reading acknowledgements for compliance with regulatory standards like ISO, SOC, FDA, AS9100, ITAR, NIST/CMMC and more using Document Locator’s windows-based document control system.
Centralized Document Control
Document Locator is a centralized platform for document control.
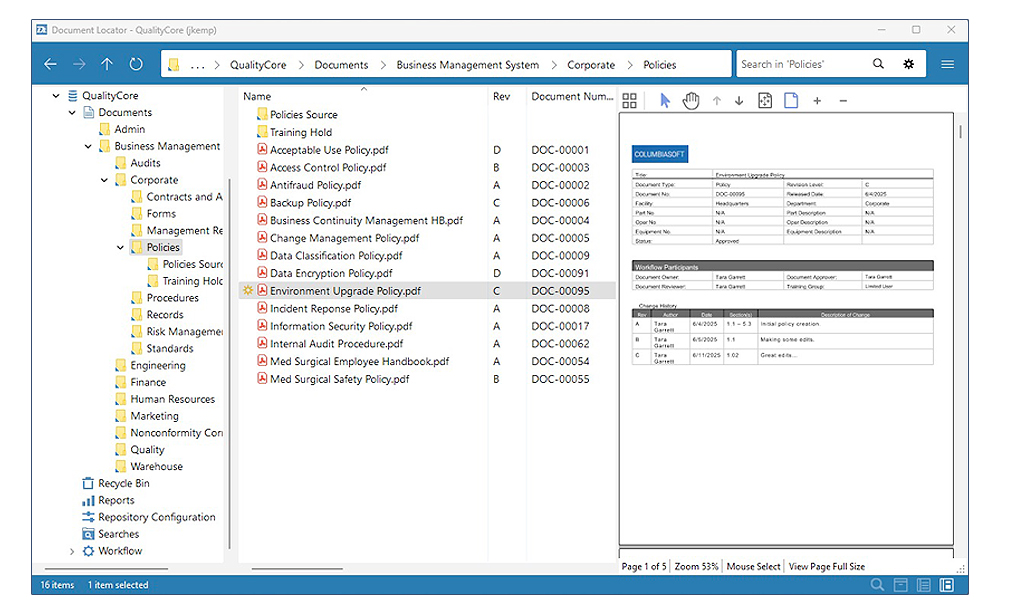
Document control is easy and intuitive with a familiar Windows-style interface. Simply right click on a document to access a menu of document control commands.
Click screenshot for larger view
Track results, not spreadsheets
Automate policy management
Improve quality & compliance
Send for required reading
Quality Documents
Quality documentation like CAPAs, SOPs, non-conformance reports, employee training, work instructions, and other programs are managed within a document control repository that provides for version tracking, approvals, notifications, policy administration, and more. Automated, defined workflows ensure the reliability and repeatability of key processes, eliminating the errors, delays, and duplications common in manual, paper-based processes.
A recent FDA audit clearly demonstrated the value of Document Locator by reducing the time it took us to retrieve requested records in half.”
Document Control Software Benefits
centralized document control system that includes version tracking, approvals, notifications, and policy administration.
How can document control streamline compliance and efficiency?
A document control system ensures that all documents are up-to-date, easily accessible, and securely maintained. This software automates the creation, review, approval, distribution, acknowledgment, and archiving of documents, reducing errors and ensuring regulatory compliance.
Document control systems enhance accountability and transparency by centralizing document storage and implementing version control to guarantee only the most current, approved documents are in use. Features such as automated audit trails and access controls provide comprehensive records of document changes and user interactions, supporting compliance initiatives and promoting a culture of continuous improvement and operational excellence.
Document Control FAQs
Document control is the practice of managing and regulating documents to ensure they are accurate, accessible, and up to date, facilitating consistency and compliance within an organization. A document control system streamlines these processes by offering a centralized platform for creating, distributing, updating, and tracking compliance with organizational guidelines and standards.
The purpose of document control is to maintain the integrity and reliability of documents, ensuring that only the most current and approved versions are in use, thereby supporting compliance, quality assurance, and effective communication.
Document version control involves tracking document changes, maintaining a history of those changes, and ensuring that only the latest version is accessible to users. Document control software can automate this process.
Improving document control processes can be achieved by implementing document control software, standardizing procedures, training employees, regularly auditing document practices, and automating workflows to enhance efficiency and accuracy.
Document control improves productivity by reducing time spent searching for documents, minimizing errors through version control, streamlining workflows, and ensuring that employees have access to accurate and up-to-date information, thereby enhancing overall operational efficiency.
Ready for a Document Control Software Demo?
Fill out the form to get started.